Quantifying Total Allowable Error Violations in Serum-Sodium Quality Control
A Computer Simulation Experiment of Two- to Six-Sigma Processes
DOI:
https://doi.org/10.21141/PJP.2025.08Keywords:
Rstudio, total allowable error, TEa, quality control, six sigma, sigma metricsAbstract
Background. Serum-sodium reporting tolerates a total allowable error (TEa) of only ±4 mmol/L, yet many laboratories continue to operate at the marginal three-sigma level because the quantitative benefit of additional sigma capability is poorly characterized.
Objectives. The study aims to translate sigma metrics into clinically intuitive risk estimates by (1) quantifying the proportion of QC results that exceed the TEa at five sigma levels (2 – 6 σ) and (2) determining whether successive sigma gains produce statistically significant reductions in error.
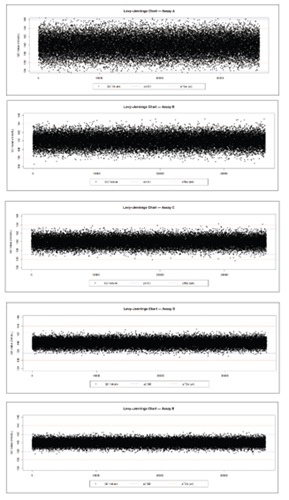
Methodology. Five (5) hypothetical assays were parameterized with a common mean of 140 mmol/L and CVs corresponding to 2-, 3-, 4-, 5- and 6-sigma performance. For each assay, 1,000 Monte-Carlo iterations were run, each iteration simulating 36,500 QC results (assuming 100 runs/day for 365 days) drawn from N(µ = 140, σ = µ × CV). The error rate (the proportion of results outside ±4 mmol/L) was recorded per iteration. Distributions were summarized (mean, range, SD); differences were evaluated with one-way ANOVA followed by Tukey’s HSD.
Results. Mean (±SD) error rates declined significantly with increasing sigma: Assay A (2σ): 0.0456 ± 0.0011; Assay B (3σ): 0.00270 ± 0.00027;Assay C (4σ): 6.3 × 10⁻⁵ ± 4.1 × 10⁻⁵; Assay D (5σ): 5.8 × 10⁻⁷ ± 8.0 × 10⁻⁷; and Assay E (6σ): 2.0 × 10⁻⁷ ± 3.1 × 10⁻⁷. The maximum single-iteration error rate fell from 0.0505 at 2 σ to 1.1 × 10⁻⁴ at 4 σ. The 5σ and 6σ processes produced zero TEa violations in ≥96 % of iterations. ANOVA confirmed a global difference (p < 0.001); all pairwise contrasts were significant (p < 0.001) except between 5 σ vs 6 σ (p = 0.62).
Conclusions. Each one-sigma gain yields an order-of-magnitude reduction in TEa violations until a plateau is reached at ≥5 σ, where residual analytical risk is negligible. These simulations support the recommendation that laboratories operating serum-sodium assays below 4 σ should prioritize precision improvements or enhanced QC strategies, whereas ≥5 σ assays may safely adopt less intensive QC without compromising patient safety.
Downloads
References
Spasovski G, Vanholder R, Allolio B, et al. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Eur J Endocrinol. 2014;170(3):G1-47. https://pubmed.ncbi.nlm.nih.gov/24569125 https://doi.org/10.1530/EJE-13-1020 DOI: https://doi.org/10.1530/EJE-13-1020
Tormey WP, Carney M, Cuesta M, Sreenan S. Reference Change values for sodium are ignored by the American and European treatment guidelines for hyponatremia. Clin Chem. 2015;61(12):1430-2. https://pubmed.ncbi.nlm.nih.gov/26152751 https://doi.org/10.1373/clinchem.2015.242602 DOI: https://doi.org/10.1373/clinchem.2015.242602
Al Dhaybi O, Bakris GL. Factitious acidosis and severe hypoalbuminemia caused by unsuspected assay interference. Clin Kidney J. 2021;14(3):1023-4. https://pubmed.ncbi.nlm.nih.gov/34840734 https://pmc.ncbi.nlm.nih.gov/articles/PMC8612138 https://doi.org/10.1093/ckj/sfaa074 DOI: https://doi.org/10.1093/ckj/sfaa074
Freitas F. Analytical quality specifications in the clinical laboratory – limiting analytical error with allowable error Limits; 2013.
Oosterhuis WP, Coskun A, Sandberg S, Theodorsson E. Performance specifications for sodium should not be based on biological variation. Clin Chim Acta. 2023;540:117221. https://pubmed.ncbi.nlm.nih.gov/36640931 https://doi.org/10.1016/j.cca.2023.117221 DOI: https://doi.org/10.1016/j.cca.2023.117221
Guo X, Zhang T, Gao X, et al. Sigma metrics for assessing the analytical quality of clinical chemistry assays: a comparison of two approaches. Biochem Medica. 2018;28(2):020708. https://pubmed.ncbi.nlm.nih.gov/30022883 https://pmc.ncbi.nlm.nih.gov/articles/PMC6039159 https://doi.org/10.11613/BM.2018.020708 DOI: https://doi.org/10.11613/BM.2018.020708
Luo Y, Yan X, Xiao Q, et al. Application of Sigma metrics in the quality control strategies of immunology and protein analytes. J Clin Lab Anal. 2021;35(11):e24041. https://pubmed.ncbi.nlm.nih.gov/34606652 https://pmc.ncbi.nlm.nih.gov/articles/PMC8605144 https://doi.org/10.1002/jcla.24041 DOI: https://doi.org/10.1002/jcla.24041
Westgard SA, Bayat H, Westgard JO. Selecting a Risk-Based SQC procedure for a HbA1c total QC plan. J Diabetes Sci Technol. 2018;12(4):780-5. https://pubmed.ncbi.nlm.nih.gov/28905657 https://pmc.ncbi.nlm.nih.gov/articles/PMC6134308 https://doi.org/10.1177/1932296817729488 DOI: https://doi.org/10.1177/1932296817729488
Westgard JO, Westgard SA. Quality control review: implementing a scientifically based quality control system. Ann Clin Biochem Int J Lab Med. 2016;53(1):32-50. https://pubmed.ncbi.nlm.nih.gov/26150675 https://doi.org/10.1177/0004563215597248 DOI: https://doi.org/10.1177/0004563215597248
Westgard JO. Managing quality vs. measuring uncertainty in the medical laboratory. CLin Chem Lab Med. 2010;48(1):31-40. https://pubmed.ncbi.nlm.nih.gov/19919330 https://doi.org/10.1515/CCLM.2010.024 DOI: https://doi.org/10.1515/CCLM.2010.024
Bozkaya G, Aksit M, Zeytinli Aksit M. Evaluation of clinical chemistry tests in emergency laboratory by sigma metrics. Turk J Biochem. 2018;43(1):9-14. https://doi.org/10.1515/tjb-2017-0143 DOI: https://doi.org/10.1515/tjb-2017-0143
Moodley N, Gounden V. Assessment of sigma metrics for routine chemistry testing in 4 laboratories in Kwa-Zulu Natal, South Africa. J Appl Lab Med. 2022;7(3):689-97. https://pubmed.ncbi.nlm.nih.gov/34636901 https://doi.org/10.1093/jalm/jfab117 DOI: https://doi.org/10.1093/jalm/jfab117
Feldhammer M, Brown M, Colby J, Bryksin J, Milstid B, Nichols JH. A survey of sigma metrics across three academic medical centers. J Appl Lab Med. 2021;6(5):1264-75. https://pubmed.ncbi.nlm.nih.gov/34060592 https://doi.org/10.1093/jalm/jfab028 DOI: https://doi.org/10.1093/jalm/jfab028
Westgard JO. Simulation and modeling for optimizing quality control and improving analytical quality assurance. Clin Chem. 1992;38(2):175-8. https://pubmed.ncbi.nlm.nih.gov/1540997 DOI: https://doi.org/10.1093/clinchem/38.2.175
Boyd JC, Bruns DE. Quality specifications for glucose meters: assessment by simulation modeling of errors in insulin dose. Clin Chem. 2001;47(2):209-14. https://pubmed.ncbi.nlm.nih.gov/11159768 DOI: https://doi.org/10.1093/clinchem/47.2.209
Boyd JC, Bruns DE. Effects of measurement frequency on analytical quality required for glucose measurements in intensive care units: assessments by simulation models. Clin Chem. 2014;60(4):644-50. https://pubmed.ncbi.nlm.nih.gov/24430017 https://doi.org/10.1373/clinchem.2013.216366 DOI: https://doi.org/10.1373/clinchem.2013.216366
Parvin CA. Quality-control (QC) performance measures and the QC planning process. Clin Chem. 1997;43(4):602-7. https://pubmed.ncbi.nlm.nih.gov/9105260 DOI: https://doi.org/10.1093/clinchem/43.4.602
Gernand W. How Reliable are critical error calculations? Clin Chem. 2006;52(5):895-6. https://pubmed.ncbi.nlm.nih.gov/16638957 https://doi.org/10.1373/clinchem.2006.067991 DOI: https://doi.org/10.1373/clinchem.2006.067991
R Core Team. R: A Language and environment for statistical computing [computer program]. Published online 2024. https://www.R-project.org/.
Posit Team. RStudio: Integrated development environment for R [computer program]. Published online 2025. http://www.posit.co/.
Microsoft Corporation. Microsoft Excel [computer program]. Published online 2025. https://www.microsoft.com.
Microsoft Corporation. Microsoft Word [computer program]. Published online 2025. https://www.microsoft.com.
OpenAI. ChatGPT [large language model]. Version: June 2025. Published online 2025. https://chat.openai.com/.
Jones GRD. Reevaluation of the power of error detection of Westgard multirules. Clin Chem. 2004;50(4):762-4. https://pubmed.ncbi.nlm.nih.gov/15044336 https://doi.org/10.1373/clinchem.2003.025585 DOI: https://doi.org/10.1373/clinchem.2003.025585
Liu Q, Bian G, Chen X, et al. Application of a six sigma model to evaluate the analytical performance of urinary biochemical analytes and design a risk‐based statistical quality control strategy for these assays: A multicenter study. J Clin Lab Anal. 2021;35(11):e24059. https://pubmed.ncbi.nlm.nih.gov/34652033 PMCID: PMC8605169 https://doi.org/10.1002/jcla.24059 DOI: https://doi.org/10.1002/jcla.24059
Weykamp C, Siebelder C. Evaluation of performance of laboratories and manufacturers within the framework of the IFCC model for quality targets of HbA1c. J Diabetes Sci Technol. 2018;12(4):747-52. https://pubmed.ncbi.nlm.nih.gov/29145735 https://pmc.ncbi.nlm.nih.gov/articles/PMC6134299 https://doi.org/10.1177/1932296817741320 DOI: https://doi.org/10.1177/1932296817741320
Badrick T, Bietenbeck A, Cervinski MA, et al. Patient-based real-time quality control: review and recommendations. Clin Chem. 2019;65(8):962-71. https://pubmed.ncbi.nlm.nih.gov/31263035 https://doi.org/10.1373/clinchem.2019.305482 DOI: https://doi.org/10.1373/clinchem.2019.305482
Taleb NN. Statistical consequences of fat tails: real world preasymptotics, epistemology, and applications; 2020. https://doi.org/10.48550/ARXIV.2001.10488
Lyon AW, Lyon ME. Evaluation of precision and bias specifications required to achieve the 2018 FDA guidance criteria for glucose meter performance using simulation Models. J Diabetes Sci Technol. 2020;14(3):513-8. https://pubmed.ncbi.nlm.nih.gov/31752535 https://pmc.ncbi.nlm.nih.gov/articles/PMC7576955 https://doi.org/10.1177/1932296819889639 DOI: https://doi.org/10.1177/1932296819889639
Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2025 Mark Angelo Ang, Karen Cybelle Sotalbo

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.









 @philippinepathologyjournal
@philippinepathologyjournal