Prevalence and Pattern of Antifungal Drug Minimum Inhibitory Concentration (MIC) of Invasive Candidiasis and its Associated Risk Factors
DOI:
https://doi.org/10.21141/PJP.2025.13Keywords:
candidiasis, antifungal agents, drug resistance, Candida, azolesAbstract
Background. Invasive candidiasis is defined by the growth of Candida species in the bloodstream or other internal organs. It is a global concern due to increasing multidrug resistance and high mortality rates. This study aimed to update prevalence data on Candida infections in the Philippines, analyzing demographic factors (age, sex), specimen sources, and associated risk factors. We compared antifungal resistance patterns against CLSI epidemiological cutoff values (ECVs) and clinical breakpoints and examined MIC variations by underlying disease to inform potential standardized empiric therapies.
Methodology. We conducted a retrospective analytical cross-sectional study (SLMC‑IERC approval, minimal risk) reviewing one year of Candida speciation and susceptibility results from January to December 2024 at a private tertiary hospital. All aseptically collected samples that tested positive for Candida species were included. Respiratory and wound specimens required a Gram stain demonstrating yeasts or hyphae prior to culture, while urine cultures were included only if they yielded ≥100,000 CFU/mL. Identification and susceptibility testing were performed using the VITEK 2 system, with results interpreted using CLSI breakpoints and ECVs.
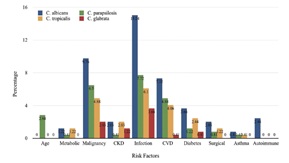
Results. Among 266 patients with Candida infections, invasive candidiasis predominated in those aged ≥ 60 years (66.4%). Candida albicans (21.7%) and C. tropicalis (13.5%) were more frequent in females, while C. parapsilosis (13.2%) and C. glabrata (5.3%) were more common in males. Blood and CSF samples strongly correlated with invasive disease and underlying risk factors. C. albicans was linked to infection-related conditions (13.9%), malignancy (9.0%), and cardiovascular disease (6.8%). C. parapsilosis (23.3%) and C. tropicalis (20.7%) were frequently associated with infection, malignancy, and metabolic disorders. C. glabrata (7.5%), noted for antifungal resistance, was isolated in patients with direct infections (3.4%) and malignancies (1.9%). Among azoles, fluconazole demonstrated greater susceptibility against Candida species, requiring lower concentrations for inhibition, despite a higher resistance rate (13.22%) compared to voriconazole (8.95%). Among echinocandins, micafungin showed better susceptibility than caspofungin. Amphotericin B demonstrated the highest overall susceptibility (93–100%), though MICs approached ECV limits. Most susceptible MIC values were: fluconazole 0.5 µg/mL for C. albicans and C. parapsilosis, 1.0 µg/mL for C. tropicalis; voriconazole and caspofungin 0.12 µg/mL; micafungin 0.06 µg/mL; amphotericin B 0.5 µg/mL; and flucytosine <1 µg/mL for all species.
Conclusion. These findings support a species-specific, risk-adapted approach to antifungal therapy, incorporating demographic and clinical variables. Continuous surveillance of invasive candidiasis prevalence and antifungal MIC trends, with periodic breakpoint updates, is crucial to preserve therapeutic efficacy. Effective management of multidrug-resistant Candida infections also requires close collaboration between clinicians and pharmacists, as well as the development of new dosing strategies based on pharmacokinetic/pharmacodynamic (PK/PD) principles.
Downloads
References
Centers for Disease Control and Prevention. Candidiasis basics. Updated April 24, 2024. Accessed April 24, 2024. https://www.cdc.gov/candidiasis/about/index.html
Oliva A, De Rosa FG, Mikulska M, et al. Invasive Candida infection: epidemiology, clinical and therapeutic aspects of an evolving disease and the role of rezafungin. Expert Rev Anti Infect Ther. 2023;21(9):957–75. https://pubmed.ncbi.nlm.nih.gov/37494128 https://doi.org/10.1080/14787210.2023.2240956 DOI: https://doi.org/10.1080/14787210.2023.2240956
Ahmed A, Azim A, Baronia AK, Marak KR, Gurjar M. Risk prediction for invasive candidiasis. Indian J Crit Care Med. 2014;18(10):682-8. https://pubmed.ncbi.nlm.nih.gov/25316979 https://pmc.ncbi.nlm.nih.gov/articles/PMC4195199 https://doi.org/10.4103/0972-5229.142178 DOI: https://doi.org/10.4103/0972-5229.142178
Soriano A, Honore PM, Puerta-Alcalde P, et al. Invasive candidiasis: current clinical challenges and unmet needs in adult populations. J Antimicrob Chemother. 2023;78(7):1569-85. https://pubmed.ncbi.nlm.nih.gov/37220664 https://pmc.ncbi.nlm.nih.gov/articles/PMC10320127 https://doi.org/10.1093/jac/dkad139 DOI: https://doi.org/10.1093/jac/dkad139
Fatahinia M, Halvaeizadeh M, Zarei Mahmoudabadi A, AboualiGalehdari E, Kiasat N. In vitro antifungal susceptibilities of six antifungal drugs against clinical Candida glabrata isolates according to EUCAST. Curr Med Mycol. 2020;6(2):1-6. https://pubmed.ncbi.nlm.nih.gov/33628974 https://pmc.ncbi.nlm.nih.gov/articles/PMC7888517 https://doi.org/10.18502/CMM.6.2.2692 DOI: https://doi.org/10.18502/cmm.6.2.2692
Juayang AC, Lim JPT, de los Reyes ZCB, et al. Antifungal resistance of Candida species in Bacolod City, Philippines. J Infect Dis Epidemiol. 2019;5:076. https://doi.org/10.23937/2474-3658/1510076 DOI: https://doi.org/10.23937/2474-3658/1510076
Dayrit M, Lagrada L, Picazo O, et al. The Philippines Health System Review. Vol 8, No. 2. World Health Organization, Regional Office for South-East Asia; 2018. Accessed September 20, 2018. https://iris.who.int/server/api/core/bitstreams/d3ecbe57-f53f-4f89-992a-3c742ad1f57a/content
Mahmoudabad AZ, Rezaei-Matehkolaei A, Ghanavati F. The susceptibility patterns of Candida species isolated from urine samples to posaconazole and caspofungin. Jundishapur J Microbiol. 2015;8(3):e24298. https://pubmed.ncbi.nlm.nih.gov/25861442 https://pmc.ncbi.nlm.nih.gov/articles/PMC4386077 https://doi.org/10.5812/jjm.24298 DOI: https://doi.org/10.5812/jjm.24298
Lamoth F, Lewis RE, Kontoyiannis DP. Role and interpretation of antifungal susceptibility testing for the management of invasive fungal infections. J Fungi (Basel). 2021;7(1):17. https://pubmed.ncbi.nlm.nih.gov/33396870 https://pmc.ncbi.nlm.nih.gov/articles/PMC7823995 https://doi.org/10.3390/jof7010017 DOI: https://doi.org/10.3390/jof7010017
Ben-Ami R. Treatment of invasive candidiasis: a narrative review. J Fungi. 2018;4(3):97. https://pubmed.ncbi.nlm.nih.gov/30115843 https://pmc.ncbi.nlm.nih.gov/articles/PMC6162658 https://doi.org/10.3390/jof4030097 DOI: https://doi.org/10.3390/jof4030097
Itable JR, Alejandria M, Mendoza M. Risk factors, species distribution and susceptibility of candidemia in the Philippines. Int J Infect Dis. 2014;21(Suppl):285. DOI: https://doi.org/10.1016/j.ijid.2014.03.1012
Binder U, Aigner M, Risslegger B, Hörtnagl C, Lass-Flörl C, Lackner M. Minimal inhibitory concentration (MIC) phenomena in Candida albicans and their impact on the diagnosis of antifungal resistance. J Fungi. 2019;5(3):83. https://pubmed.ncbi.nlm.nih.gov/31487830 https://pmc.ncbi.nlm.nih.gov/articles/PMC6787722 https://doi.org/10.3390/jof5030083 DOI: https://doi.org/10.3390/jof5030083
Kaur R, Dhakad MS, Goyal R, Haque A, Mukhopadhyay G. Identification and antifungal susceptibility testing of Candida species: a comparison of Vitek 2 system with conventional and molecular methods. J Glob Infect Dis. 2016;8(4):139-46. https://pubmed.ncbi.nlm.nih.gov/27942193 https://pmc.ncbi.nlm.nih.gov/articles/PMC5126752 https://doi.org/10.4103/0974-777X.192969 DOI: https://doi.org/10.4103/0974-777X.192969
Barantsevich N, Barantsevich E. Diagnosis and treatment of invasive candidiasis. Antibiotics (Basel). 2022;11(6):718. https://pubmed.ncbi.nlm.nih.gov/35740125 https://pmc.ncbi.nlm.nih.gov/articles/PMC9219674 https://doi.org/10.3390/antibiotics11060718 DOI: https://doi.org/10.3390/antibiotics11060718
Meena DS, Kumar D. Candida pneumonia: an innocent bystander or a silent killer? Med Princ Pract. 2022;31(1):98-102. https://pubmed.ncbi.nlm.nih.gov/34638123 https://pmc.ncbi.nlm.nih.gov/articles/PMC8995637 https://doi.org/10.1159/000520111 DOI: https://doi.org/10.1159/000520111
Korem M, Geffen Y, Amit S. Don’t mess with the machine—evaluation of fluconazole susceptibility testing for Candida glabrata using the new VITEK 2 AST-YS08 card following species modification. Diagn Microbiol Infect Dis. 2020;96(2):114896. https://pubmed.ncbi.nlm.nih.gov/31780195 https://doi.org/10.1016/j.diagmicrobio.2019.114896 DOI: https://doi.org/10.1016/j.diagmicrobio.2019.114896
Carvalhaes C. When should antifungal susceptibility testing be performed for Candida species isolated from clinical specimens? AST News Update 2019: Practical Tips 1 – AST for Candida species. 2019. https://clsi.org/about/news/ast-news-update-2019-practical-tips-1/
Yamin D, Wakil AM, Goni MD, Irekeola AA, Hajissa K. Trends of Candida species causing bloodstream infections in South-Eastern Asia: a systematic review and meta-analysis. Asian Pac J Trop Med. 2025;18(2):51-66. DOI: 10.4103/apjtm.apjtm_559_24. DOI: https://doi.org/10.4103/apjtm.apjtm_559_24
Franconi I, Rizzato C, Poma N, Tavanti A, Lupetti A. Candida parapsilosis sensu stricto antifungal resistance mechanisms and associated epidemiology. J Fungi (Basel). 2023;9(8):798. https://pubmed.ncbi.nlm.nih.gov/37623569 https://pmc.ncbi.nlm.nih.gov/articles/PMC10456088 https://doi.org/10.3390/jof9080798 DOI: https://doi.org/10.3390/jof9080798
Pappas PG, Kauffman CA, Andes DR, et al. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis. 2016;62(4):e1-50. Phttps://pubmed.ncbi.nlm.nih.gov/26679628 https://pmc.ncbi.nlm.nih.gov/articles/PMC4725385 https://doi.org/10.1093/cid/civ933 DOI: https://doi.org/10.1093/cid/civ933
Pea F, Lewis RE. Overview of antifungal dosing in invasive candidiasis. J Antimicrob Chemother. 2018;73(Suppl 1):i33-43. https://pubmed.ncbi.nlm.nih.gov/29304210 https://doi.org/10.1093/jac/dkx447 DOI: https://doi.org/10.1093/jac/dkx447
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Angeli Joana Robillos, Evelina Lagamayo

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.









 @philippinepathologyjournal
@philippinepathologyjournal